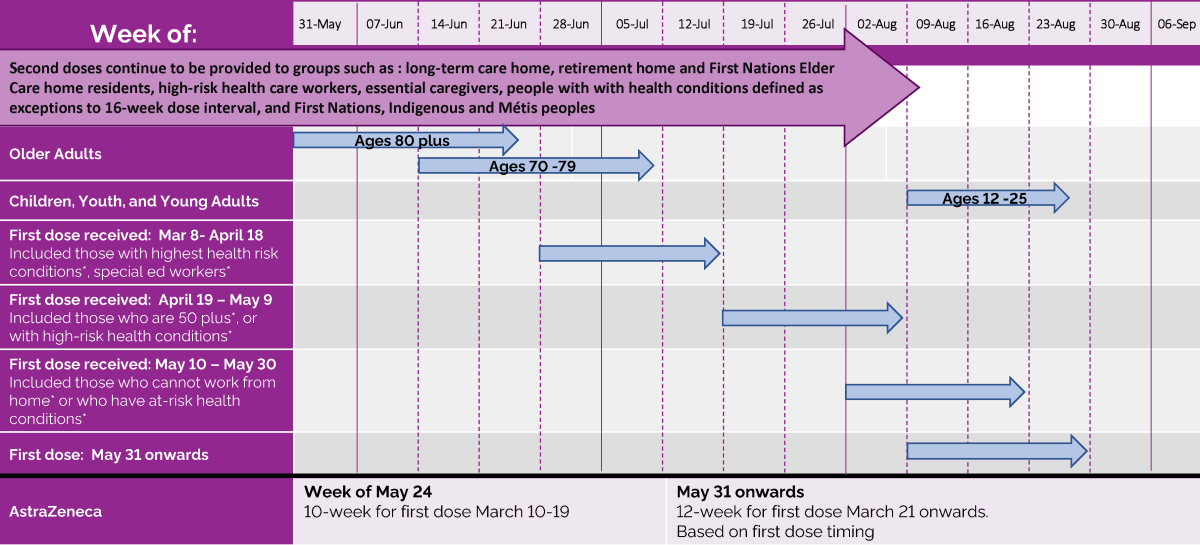
Using recent estimates, we parameterized the infectivity of asymptomatic, mild symptomatic, and severe symptomatic individuals to be 26%, 44%, and 89% relative to the pre-symptomatic stage. In our agent-based model, the risk of infection for people susceptible to COVID-19 depended on contact with infectious individuals that could be in asymptomatic, pre-symptomatic, or symptomatic stages of the disease. A DSD strategy with Pfizer-BioNTech vaccines is comparatively inferior to Moderna vaccines, and the delay to achieve maximum benefits depends on the durability of the first-dose efficacy. The maximum benefits would be achieved with a delay of at least 9 weeks from the recommended schedule for administering the second dose. For Moderna’s two-dose vaccine, we show that a DSD strategy would outperform the recommended interval between doses in terms of reducing the number of hospitalizations and deaths. We determined the optimal timing for administering the second dose based on vaccine efficacy estimated in clinical trials and population-level studies following first and second doses. Here, we employed an agent-based model of COVID-19 transmission and vaccination to compare the epidemiological impact of tested and DSD vaccination schedules, considering a range of pre-existing immunity accrued since the emergence of COVID-19. However, the conditions under which this improvement is achievable remain unexamined, such as the durability of first-dose efficacy and protection against infection. Broader population-level protection against COVID-19 in a delayed second dose (DSD) strategy, even with lower individual-level efficacy from the first dose in the short term, may improve the impact of vaccination compared to the recommended two-dose strategy that provides more complete protection to a smaller subset of the population. The emergence of novel, more contagious SARS-CoV-2 variants in several countries, and the potential for their widespread transmission have led to a public health conundrum regarding whether to vaccinate more individuals with the first dose of available vaccines and delay the second dose, or to prioritize completion of the two-dose series based on tested schedules in clinical trials. In contrast to the remarkable speed of development, vaccine delivery has proven to be challenging due to supply shortages and limited distribution capacity in several countries. Clinical trials and evaluations of mass vaccination campaigns have demonstrated that these vaccines can provide high levels of protection against symptomatic and severe disease with two doses administered three to four weeks apart. A number of vaccines, including those developed by Pfizer-BioNTech, Moderna, and Oxford-AstraZeneca, have received emergency use authorization from regulatory bodies in different countries.  
The spread of coronavirus disease 2019 (COVID-19) has ravaged global health and suppressed economic activity despite the range of mitigation measures implemented by countries worldwide. 
Our findings underscore the importance of quantifying the characteristics and durability of vaccine-induced protection after the first dose in order to determine the optimal time interval between the two doses. However, there was no clear advantage of delaying the second dose with Pfizer-BioNTech vaccines in reducing infections, unless the efficacy of the first dose did not wane over time. Pfizer-BioNTech vaccines also averted an additional 0.61 (95% CrI: 0.37 – 0.89) hospitalizations and 0.31 (95% CrI: 0.23 – 0.45) deaths per 10,000 population in a 9-week delayed second dose strategy compared to the 3-week recommended schedule between doses. Our results show that for Moderna vaccines, a delay of at least 9 weeks could maximize vaccination program effectiveness and avert at least an additional 17.3 (95% CrI: 7.8 – 29.7) infections, 0.71 (95% CrI: 0.52 – 0.97) hospitalizations, and 0.34 (95% CrI: 0.25 – 0.44) deaths per 10,000 population compared to the recommended 4-week interval between the two doses. We developed an agent-based model of COVID-19 transmission to compare the impact of these two vaccination strategies, while varying the temporal waning of vaccine efficacy following the first dose and the level of pre-existing immunity in the population. Constraints in vaccine supply and distribution capacity, together with a deadly wave of COVID-19 from November 2020 to January 2021 and the emergence of highly contagious SARS-CoV-2 variants, sparked a policy debate on whether to vaccinate more individuals with the first dose of available vaccines and delay the second dose, or to continue with the recommended two-dose series as tested in clinical trials. Two of the COVID-19 vaccines currently approved in the United States require two doses, administered three to four weeks apart.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
